Polyethylene: The Accidental British Invention That Changed the World
Published on: 30/04/2026
 A chemist at work in an industrial laboratory of the era — the kind of setting where the polyethylene story began.
A chemist at work in an industrial laboratory of the era — the kind of setting where the polyethylene story began.
In March 1933, two chemists at an ICI laboratory in Cheshire were trying to make ethylene react with benzaldehyde under crushing pressure — around 1,400 atmospheres. The experiment failed. But when Eric Fawcett and Reginald Gibson opened the steel reaction vessel, they found something unexpected coating its walls: a waxy white solid.
What they had created, entirely by accident, was the world’s first usable polyethylene — the material that would go on to become the most widely produced plastic on Earth.
A Happy Accident in Cheshire
The discovery shouldn’t really have worked at all. Fawcett and Gibson’s reaction vessel had a tiny leak, and a trace of oxygen had crept in. That oxygen acted as an unintentional catalyst, kicking the ethylene molecules into bonding together in long, repeating chains — a process now known as polymerisation.
There had been hints of polyethylene before. The German chemist Hans von Pechmann had produced a similar waxy substance back in 1898 and called it polymethylene, though he had no way of recognising what he had found. The Cheshire team came closer, but their early results were maddeningly inconsistent. Without knowing the leak had been the magic ingredient, they couldn’t reproduce it reliably for nearly two years.
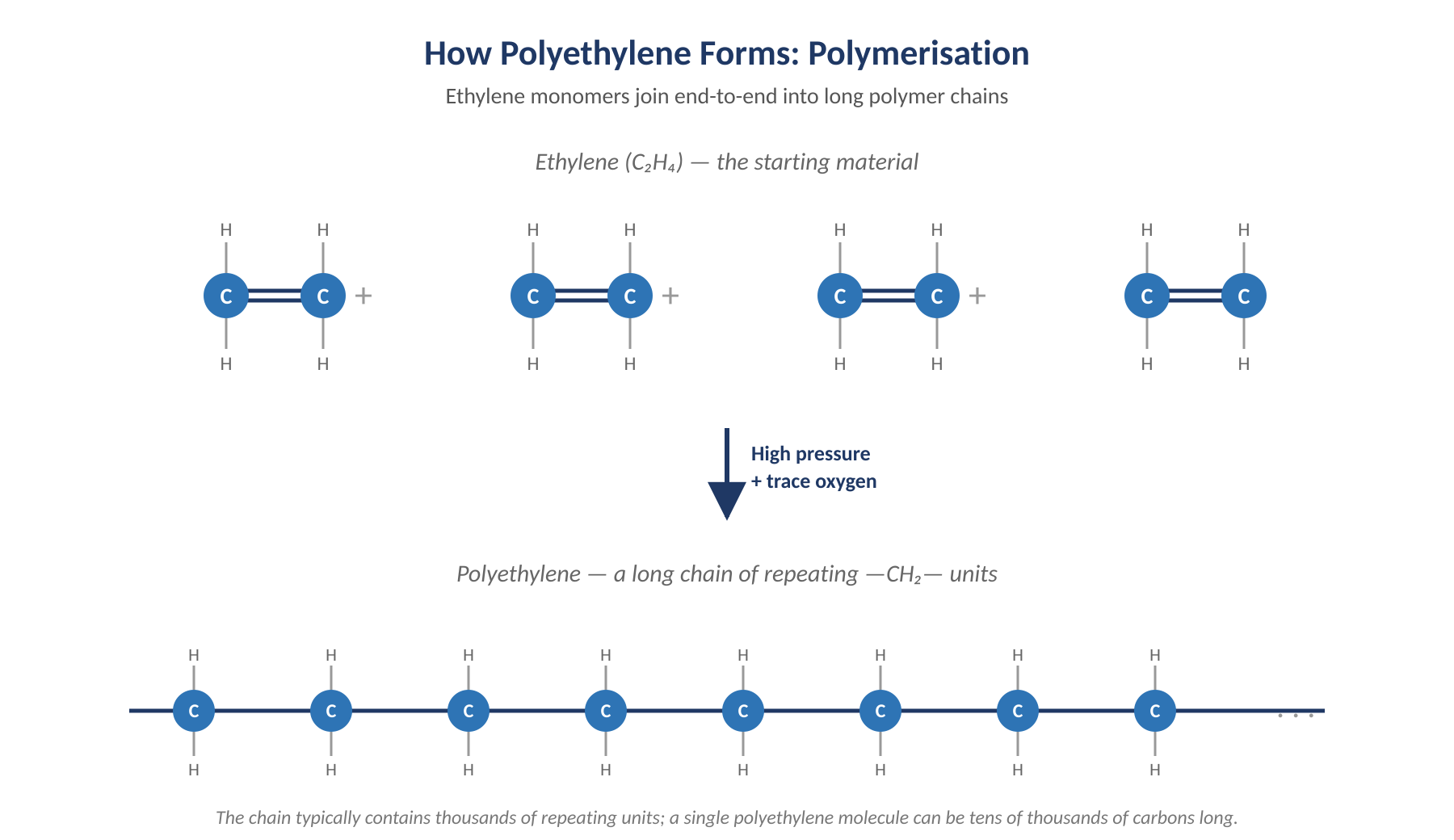 Ethylene monomers join end-to-end into long polyethylene chains.
Ethylene monomers join end-to-end into long polyethylene chains.
The breakthrough came in 1935, when fellow ICI chemist Michael Perrin worked out a controlled high-pressure method that introduced just the right amount of oxygen on purpose. With that, polyethylene could finally be made on demand — and a laboratory curiosity began its journey towards becoming an industrial workhorse.
A Wartime Secret
By 1940, polyethylene was classified as a strategic material. Its combination of light weight, moisture resistance, and exceptional electrical insulation made it invaluable to the war effort, and Britain treated it as a closely guarded military secret.
Its most important wartime role was in radar. Polyethylene’s low signal loss at high frequencies made it a near-perfect insulator for radar equipment — and crucially, it was light enough to carry into the air. Airborne radar, fitted into aircraft to spot enemy planes and U-boats, owes a great deal to a material that was, just a few years earlier, an unintended residue in a leaky steel cylinder.
 An Avro Lancaster in flight during WWII. Many Lancasters were fitted with the H2S airborne radar — a system whose effectiveness relied on polyethylene's exceptional electrical insulating properties.
An Avro Lancaster in flight during WWII. Many Lancasters were fitted with the H2S airborne radar — a system whose effectiveness relied on polyethylene's exceptional electrical insulating properties.
Out of the Lab and Into Everything
After the war, the secret was out, and polyethylene moved quickly into commercial production. New catalysts developed in the late 1940s and 1950s — particularly the Ziegler–Natta process — allowed it to be made at much lower temperatures and pressures. The material rapidly became one of the fastest-growing products in the plastics industry, with ICI leading UK manufacturing and exporting both the material and the technology across Europe.
Today, polyethylene accounts for roughly a third of all plastic produced globally, with more than 100 million tonnes manufactured each year. Packaging takes the largest share — around 40% — but its strength, chemical resistance, and machinability mean it also turns up in water and gas pipes, electrical cable insulation, and engineered components where wear resistance matters.
In the UK alone, the polyethylene market was valued at around £2.15 billion in 2024, accounting for roughly 1.8% of the global total (Grand View Research). The wider UK plastics industry processes around 3.5 million tonnes of plastic material every year (British Plastics Federation).
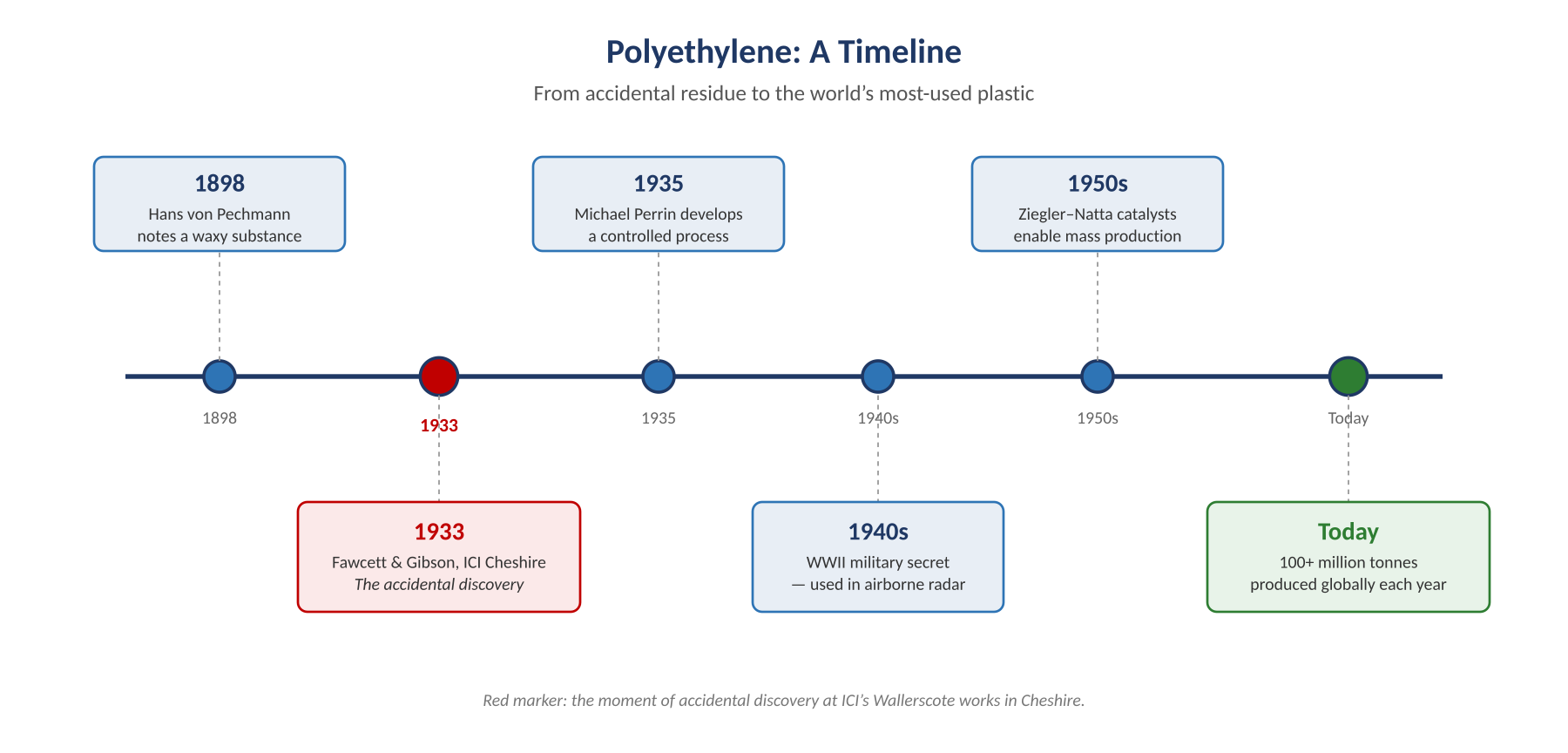 A timeline of polyethylene — from accidental residue to global commodity.
A timeline of polyethylene — from accidental residue to global commodity.
What Next for Polyethylene?
The same durability that makes polyethylene useful also makes it persistent, and global recycling rates remain low despite HDPE being technically straightforward to reprocess. The industry's response is gradual rather than revolutionary — improved sorting and reprocessing on one side, and the rise of bio-based polyethylene, made from sugarcane ethanol rather than fossil feedstocks, on the other. The likelier future is not the disappearance of polyethylene, but a quieter evolution of the same accidental discovery that began in a Cheshire laboratory in 1933.
Polyethylene at Simply Plastics
We stock polyethylene in our engineering plastics range, available as HDPE and HMWPE sheet and rod in a range of colours. You can order yours here, or get in touch at [email protected] if you’d like advice on the right grade for your application.
Frequently asked questions
Yes, both PE300 and PE500 are waterproof and have low moisture absorption.
Polyethylene is difficult to bond because it has a low surface energy. However, it can be bonded with special adhesives and methods

 {{product.colours}} Colours
{{product.colours}} Colours